Late-Stage Derivatization of Oleanolic Acid-Based Anti-HIV-1 Compounds
Reon Takeuchi, Junko Fujimoto, Yoshinori Taguchi, Ryuji Ide, Ryuji Kyan, Kohei Sato, Nobuyuki Mase, Masaru Yokoyama, Shigeyoshi Harada, Tetsuo Narumi
Chem. Pharm. Bull. 2024, 72, 330-335.
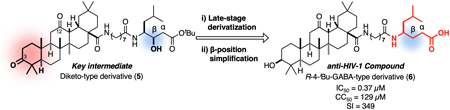
A 12-keto-type oleanolic acid derivative (4) has been identified as a potent anti-human immunodeficiency virus type-1 (HIV-1) compound that demonstrates synergistic effects with several types of HIV-1 neutralizing antibodies. In the present study, we used a common key synthetic intermediate to carry out the late-stage derivatization of an anti-HIV compound based on the chemical structure of a 12-keto-type oleanolic acid derivative. To execute this strategy, we designed a diketo-type oleanolic acid derivative (5) for chemoselective transformation, targeting the carboxy group and the hydroxyl group on the statine unit, as well as the 3-carbonyl group on the oleanolic acid unit, as orthogonal synthetic handles. We carried out four types of chemoselective transformations, leading to identification of the indole-type derivative (16) as a novel potent anti-HIV compound. In addition, further optimization of the β-hydroxyl group on the statine unit provided the R-4-isobutyl γ-amino acid-type derivative (6), which exhibited potent anti-HIV activity comparable to that of 4 but with reduced cytotoxicity.